کتاب THE MERCK INDEX به صورت آنلاین
یکی از کتاب های بسیار مفید و کاربردی در علم شیمی کتاب معروف THE MERCK INDEX میباشد.در این کتاب اطلاعات بسیار ارزشمندی در مورد ترکیبات شیمیایی ساخته شده و سنتز شده وجود دارد که برای شیمیدان ها و افرادی که با ترکیبات شیمیایی سر و کار دارند ، بسیار مفید است.این کتاب ارزشمند به صورت آنلاین بر روی وبلاگ شیمی 4800 قرار داده شده است تا دوستان از آن استفاده کنند.
کتاب THE MERCK INDEX به صورت آنلاین
استفاده از این مطلب با لینک به این وبلاگ مجاز است.
اطلاعات مربوط به ترکیب بنزن
تمامی اطلاعات نظیر فرمول مولکولی ، جرم مولکولی ، ساختار ترکیب ، اسامی دیگر ترکیب ، اطلاعات مربوط به سنتز ترکیب ، مقالات و پتنت های سنتز و تهیه این ترکیب ، نقطه جوش ، نقطه ذوب ، دانسیته و ... را به شما خواهد داد.
یکی از کتاب های بسیار مفید و کاربردی در علم شیمی کتاب معروف THE MERCK INDEX میباشد.در این کتاب اطلاعات بسیار ارزشمندی در مورد ترکیبات شیمیایی ساخته شده و سنتز شده وجود دارد که برای شیمیدان ها و افرادی که با ترکیبات شیمیایی سر و کار دارند ، بسیار مفید است.این کتاب ارزشمند به صورت آنلاین بر روی وبلاگ شیمی 4800 قرار داده شده است تا دوستان از آن استفاده کنند.
کتاب THE MERCK INDEX به صورت آنلاین
استفاده از این مطلب با لینک به این وبلاگ مجاز است.
-------------------------------------------------------------------------------------------------------------------------------
مثال :اطلاعات مربوط به ترکیب بنزن
تمامی اطلاعات نظیر فرمول مولکولی ، جرم مولکولی ، ساختار ترکیب ، اسامی دیگر ترکیب ، اطلاعات مربوط به سنتز ترکیب ، مقالات و پتنت های سنتز و تهیه این ترکیب ، نقطه جوش ، نقطه ذوب ، دانسیته و ... را به شما خواهد داد.
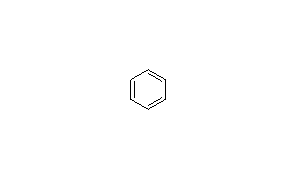
|
| |
Title: Benzene CAS Registry Number: 71-43-2 Additional Names: Benzol; cyclohexatriene Molecular Formula: C6H6 Molecular Weight: 78.11 Percent Composition: C 92.26%, H 7.74% Literature References: Natural component of petroleum, usually <1.0% by weight. Discovered by Faraday in compressed oil gas in 1825. Obtained in the coking of coal and in the production of illuminating gas from coal. Manuf by catalytic reforming and separation of aromatic compounds, thermal or catalytic dealkylation of toluene, toluene disproportionation; from pyrolysis gasoline. Purification by washing with water: GB 863711 (1961 to Schloven-Chemie and H. Koppers GmbH), C.A. 55, 16971f (1961). Lab prepn from aniline: Gattermann-Wieland, Praxis des Organischen Chemikers (de Gruyter, Berlin, 40th ed., 1961) p 247. Production of pure benzene: French, Ind. Chem. 39, 9-12 (1963). Manuf: Faith, Keyes & Clark's Industrial Chemicals, F. A. Lowenheim, M. K. Moran, Eds. (Wiley-Interscience, New York, 4th ed., 1975) pp 126-137. Physical properties: Thorne et al., Ind. Eng. Chem. Anal. Ed. 17, 481 (1945). Solubility studies: F. P. Schwarz, Anal. Chem. 52, 10 (1980). Toxicity data: Kimura et al., Toxicol. Appl. Pharmacol. 19, 699 (1971). Review: W. Fruscella in Kirk-Othmer Encyclopedia of Chemical Technology vol. 4 (Wiley-Interscience, New York, 4th ed., 1992) pp 73-103. Reviews of toxicology: R. Snyder et al., Rev. Biochem. Toxicol. 3, 123-154 (1981); D. J. Paustenbach et al., Environ. Health Perspect. 101, Suppl. 6, 177-200 (1993); and human exposure: Toxicological Profile for Benzene (PB98-101157, 1997) 463 pp. Symposia on metabolism, toxicity and carcinogenesis: Environ. Health Perspect. 82, 3-310 (1989); ibid. 104, Suppl. 6, 1121-1441 (1996). Properties: Clear, colorless, volatile, highly flammable liquid; characteristic odor. d415 0.8787. bp 80.1°. mp 5.5°. nD20 1.50108. Flash pt, closed cup: 12°F (-11°C). Soly in water at 23.5°C (w/w): 0.188%. Miscible with alcohol, chloroform, ether, carbon disulfide, carbon tetrachloride, glacial acetic acid, acetone, oils. Keep in well-closed containers in a cool place and away from fire. LD50 orally in young adult rats: 3.8 ml/kg (Kimura). Melting point: mp 5.5° Boiling point: bp 80.1° Flash point: Flash pt, closed cup: 12°F (-11°C) Index of refraction: nD20 1.50108 Density: d415 0.8787 Toxicity data: LD50 orally in young adult rats: 3.8 ml/kg (Kimura) Derivative Type: Sodium deriv Additional Names: Phenyl sodium Molecular Formula: C6H5Na Molecular Weight: 100.09 Percent Composition: C 72.00%, H 5.04%, Na 22.97% Literature References: Prepn: Schlosser, Angew. Chem. 76, 267 (1964). Properties: Solid mass, dec by water, acids, alkalies. Sol in liquid ammonia, tetrahydrofuran. CAUTION: Potential symptoms of overexposure by inhalation or ingestion are dizziness, headache, vomiting, visual disturbances, staggering gait, hilarity, fatigue, anorexia, lassitude, CNS depression, loss of consciousness, respiratory arrest. Chronic exposure has been associated with bone marrow depression and leukemia. Direct contact may cause irritation of eyes, nose, respiratory system and skin; dermititis may develop due to defatting action. Aspiration into the lung may lead to chemical pneumonitis. See Patty's Industrial Hygiene and Toxicology vol. 2B, G. D. Clayton, F. E. Clayton, Eds. (Wiley-Interscience, New York, 4th ed., 1994) pp 1306-1326; NIOSH Pocket Guide to Chemical Hazards (DHHS/NIOSH 97-140, 1997) p 26. Benzene is listed as a known human carcinogen: Report on Carcinogens, Eleventh Edition (PB2005-104914, 2004) p III-26. Use: Manuf of industrial chemicals such as polymers, detergents, pesticides pharmaceuticals, dyes, plastics, resins. Solvent for waxes, resins, oils, natural rubber, etc. Gasoline additive. Therap-Cat-Vet: Has been used as a disinfectant |


